|
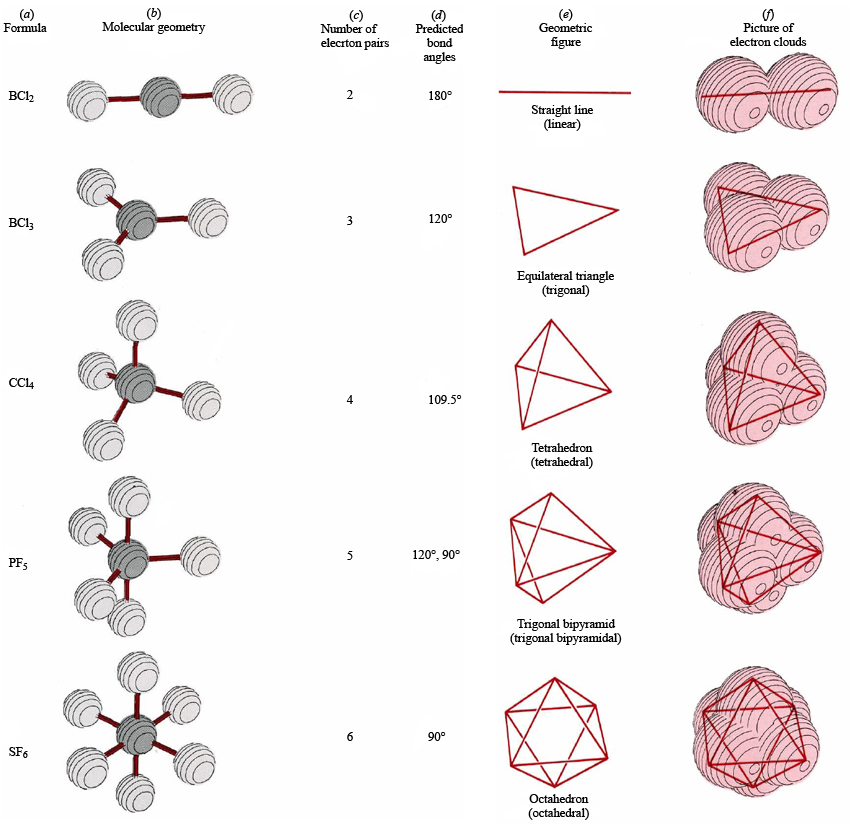
This is not needed except when looking at the. The greater the repulsion force between two pairs of electrons, the more far apart the two pairs of electrons will be and hence the larger the bond angle between them.\) illustrates several representations of the water, ammonia, and methane molecules. We can then do the same for ALL the 7 molecules in the magic list above to form a table: Shape Name Bonding Pairs Lone Pairs Total Linear 2 0 2 Trigonal Planar 3 0 3 Tetrahedral 4 0 4 Pyramidal 3 1 4 Bent 2 2 4 Trigonal Bipyramidal 5 0 5 Octahedral 6 0 6 I put a total column in the table. The bond angle between two lone pairs of electrons is the greatest.This means that the repelling effect of lone pairs is a lot stronger than that of bonding pairs. Their electron charge clouds are more concentrated and wider. Lone pairs of electrons are held a lot closer to the central atom than bonding pairs of electrons are. Lone pair electron charge clouds repel more than bonding pair electron charge clouds.Remember that like charges repel so electron charge cloud will repel each other. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. Electron charge clouds are regions of negative charge where electron pairs are found. Electron charge clouds are as far apart as possible to minimise repulsion.It does not matter whether the electron pair is involved in a bond or not. Any electron pair in the outermost shell of an atom can exist as an electron charge cloud. An electron charge cloud can contain either bonding pairs or lone pairs of electrons.An electron charge cloud is a region of negative charge around the nucleus and it is linked to an atomic orbital. An electron charge cloud is a region where there is high chance of an electron pair being present.A bonding pair of electrons is a pair of electrons that are shared between two atoms in a bond.īonding – Molecular Shapes (A-Level Chemistry) A lone pair of electrons is a pair of electrons that are not shared with another atom in a bond. The electron pairs found around the central atom can either be lone pairs or bondings pairs. The electron pairs can either be lone pairs or bonding pairs.The number of electron pairs found in the outermost shell of a central atom determines the shape of a molecule or ion. Compare bond angle predictions from the VSEPR-based model to real molecules. Name molecule and electron geometries for molecules with up to six electron groups surrounding a central atom. Recognize the difference between electron and molecular geometry. The shape of a molecule is determined by electron pairs around a central atom. Recognize that molecule geometry is due to repulsions between electron groups.The central atom forms covalent bonds with all the other atoms involved in the molecule The central atom is the atom that every other atom is bonded to.Chemistry Teacher Resource Edexcel | AQA | OCRīonding - Molecular Shapes (A-Level Chemistry) Molecular Shapes Electron Pairs.Once again, both groups around the central atom are bonding pairs (BP), so CO 2 is designated as AX 2. Like BeH 2, the arrangement that minimizes repulsions places the groups 180° apart. Biology Teacher Resource Edexcel | AQA | OCR | CIE Each double bond is a group, so there are two electron groups around the central atom.Booking A-Levels as a Private Candidate.Chemistry Flashcards Edexcel | AQA | OCR | CIE.Biology Flashcards Edexcel | AQA | OCR | CIE.Chemistry Textbook Edexcel | AQA | OCR | CIE.Biology Textbook Edexcel | AQA | OCR | CIE.Physics Weekly Classes Edexcel | AQA | OCR | CIE.Chemistry Weekly Classes Edexcel | AQA | OCR | CIE.Biology Weekly Classes Edexcel | AQA | OCR | CIE.GCSE Physics Flashcards Edexcel | AQA | OCR | CIE.Physics Online Course Edexcel | AQA | OCR | CIE.The molecular geometry is the shape of the molecule. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound. Chemistry Online Course Edexcel | AQA | OCR | CIE Molecular geometry is the name of the geometry used to describe the shape of a molecule.Biology Online Course Edexcel | AQA | OCR | CIE.Physics Tutor Edexcel | AQA | OCR | CIE Molecular geometry refers to the arrangement of atoms in a molecule in a two- or three-dimensional structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
